The CRISPR/Cas gene editing system has a lot of buzz behind it: an amusingly crunchy name, an intriguing origin, and potential uses both in research labs and even in the clinic. We heard that Emory scientists are testing it, so an explainer was in order.
The CRISPR (Clustered Regularly Interspaced Short Palindromic Repeats) system was originally discovered by dairy industry researchers seeking to prevent phages, the viruses that infect bacteria, from ruining the cultures used to make cheese and yogurt. Bacteria incorporate small bits of DNA from phages into their CRISPR region and use that information to fight off the phages by chewing up their DNA.
At Emory, infectious disease specialist David Weiss has published research on CRISPR in some types of pathogenic bacteria, showing that they need parts of the CRISPR system to evade their hosts and stay infectious. Biologist Bruce Levin has modeled CRISPR-mediated immunity’s role in bacterial evolution.
What has attracted considerable attention recently is CRISPR/Cas-derived technology, which offers the ability to dive into the genome and make a very precise change. Scientists have figured out how to retool the CRISPR/Cas machinery – the enzymes that do the chewing of the phage DNA — into enzymes that can be targeted by an external guide.
For biologists in the laboratory, this is a way to probe a gene’s function by making an animal with its genes altered in a certain way. The method is gaining popularity here at Emory. Geneticist Peng Jin reports:
“CRISPR is much more efficient and quicker than traditional homologous recombination. One can directly inject the plasmid and guide RNA into mouse embryo to make knockout mice. You can also target multiple genes at the same time.â€
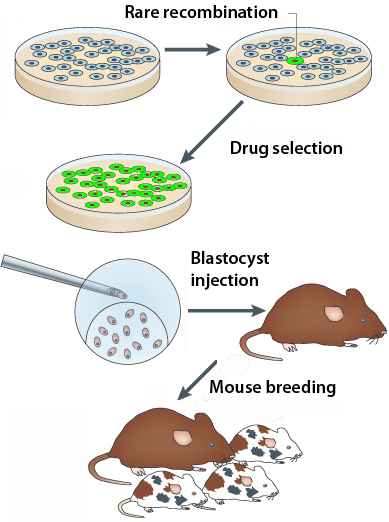
The traditional method Jin refers to involves taking cultured embryonic stem cells, zapping DNA carrying a modified or disabled gene into them, and hoping that the cells’ repair machinery sews the DNA into the genome in the right way. Usually they have to use antibiotics and drugs to screen out all the cells where the DNA gets jammed into the genome haphazardly. Also, Jin adds that CRISPR/Cas technology can be used for whole-genome screens.
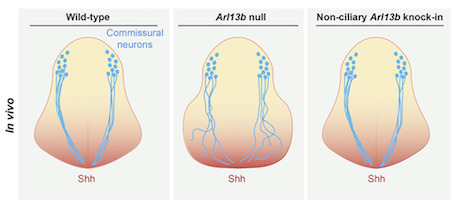
Tamara Caspary, a developmental biologist and scientific director of Emory’s transgenic mouse and gene targeting core, says she and her core team are in the process of developing and validating CRISPR, so that the technique could be accessible to many Emory investigators.
Potential clinical uses: Japanese scientists have proposed that CRISPR/Cas be employed against HIV infection. One can envision similar gene therapy applications.