In 2018, Winship magazine had a feature story on pancreatic cancer. Our team developed an illustration that we hoped could convey the tumors’ complex structure, which contributes to making them difficult to treat. Oncologist Bassel El-Rayes described how the tumors recruit other cells to form a protective shell.
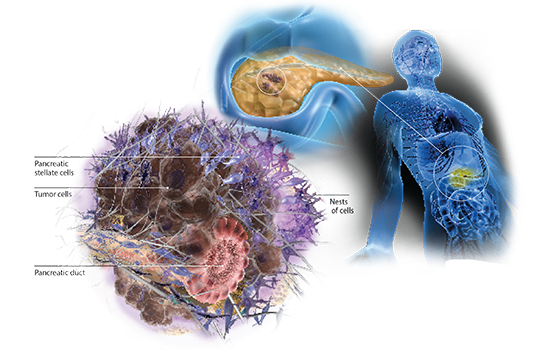
“If you look at a tumor from the pancreas, you will see small nests of cells embedded in scar tissue,” he says. “The cancer uses this scar tissue as a shield, to its own advantage.”
With El-Rayes and fellow oncologist Walid Shaib, Greg Lesinski’s lab recently published a paper in JCI Insight. The point of the paper was to look at how chemotherapy changes immune activity in the tumor microenvironment, but we also get vivid images giving us a glimpse of those nests. It helps to view these images as large as possible, so please check them out at the journal’s site, which has no paywall.

Regions stained green are tumor-rich; red regions are immune cell-rich, and blue regions are rich in stromal cells (stellate/fibroblast cells). The goal is to get immune cells to envelop the tumors more, like in square 8.
The 2018 magazine story also laid out some of Lesinski’s and El-Rayes’ ideas.
Based on his lab’s recent success in animal models, Lesinski thinks that combining an immunotherapy drug with agents that stop IL-6 could pry open pancreatic cancers’ protective shells. In those experiments, the combination resulted in fewer stellate cells and more T cells in the tumors. Fortunately, a couple of “off-the-shelf” options, drugs approved for rheumatoid arthritis, already exist for targeting IL-6, Lesinski says.
On that theme, we noticed that a clinical trial was posted on clinicaltrials.gov in December that implements those proposals: “Siltuximab and Spartalizumab in Patients With Metastatic Pancreatic Cancer”. El-Rayes is the principal investigator, and it is not yet recruiting. Siltuximab is an antibody against IL-6 and spartalizumab is a second generation PD-1 inhibitor.
Update: The XL888 + pembrolizumab study mentioned in the article is also moving along, presented by Mehmet Akce at the Gastrointestinal Cancers Symposium.





