Bypassing stem cells, Emory scientists can now create engineered heart tissue by directly reprogramming connective tissue cells in mice. The findings could provide new avenues for a quest many cardiologists have pursued: repairing the damaged heart like patching a roof.
The results were published in Nature Biomedical Engineering.
“This is the first study demonstrating direct tissue reprogramming from single adult cells from the body,” says senior author Young-sup Yoon, MD, PhD, professor of medicine at Emory University School of Medicine.
The research could potentially provide therapeutic options for millions of people with heart failure or other conditions. If heart muscle is damaged by a heart attack, the damaged or dead cells do not regenerate. Other scientists have shown they can create human heart tissue from induced pluripotent stem cells (example), but the Emory team showed that it is possible to avoid stem cells and the technologies required to create them, such as viruses.
“Direct reprogramming into tissues that contain multiple cell types has not previously been reported, and it could open new pathways in the regenerative medicine field,” Yoon says. “It could serve as a platform for cell-based therapy by avoiding the problems of current stem cell-based approaches, and for disease modeling and drug development.”
Yoon is also part of the Wallace H. Coulter Department of Biomedical Engineering at Georgia Tech and Emory. First author Jaeyeaon Cho, PhD was a post-doctoral fellow at Emory and is currently a research assistant professor at Yonsei University College of Medicine in South Korea. Emory faculty members Rebecca Levit, MD and Hee Cheol Cho, PhD are co-authors on the paper.
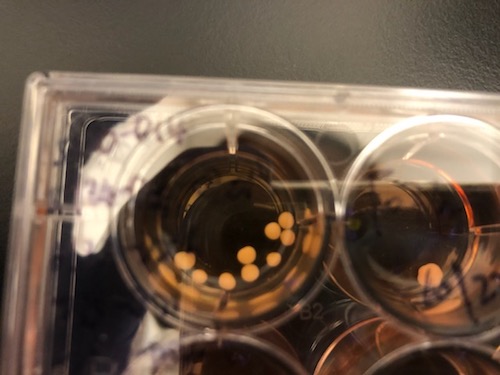
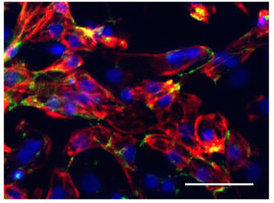
Applying a combination of growth factors, regulatory microRNA and vitamins, the Emory researchers could create tissue that contains cardiac muscle, along with blood vessels containing endothelial cells and smooth muscle cells, and fibroblasts. In culture, the four cell types weave themselves together, bypassing any need to build heart tissue from separate components.
When transplanted onto the damaged heart of a mouse after a simulated heart attack, cells from the engineered tissue can migrate into the host heart, and improve its functioning.
“In some previous studies, when a tissue patch composed of engineered cells and supportive biomaterials was transplanted to the damaged heart, there was little or no migration of cells from the patch to the host heart,” Yoon says.

The critical elements of the direct reprogramming approach are microRNAs, which are “master keys” that control several genes at once. The researchers discovered the potential of one microRNA fortuitously; a pilot study examined the effect of applying several microRNAs active in the heart to fibroblasts. Unexpectedly, one of them generated endothelial cell and smooth muscle along with cardiac muscle cells.
The Emory researchers say that their engineered tissue does not exactly mimic natural heart tissue. The cardiac muscle cells do spontaneously contract, but they display immature characteristics. But after transplantation, the engrafted cells mature and integrate into the host heart. Over 16 weeks, the engrafted cells become indistinguishable from the host cardiac muscle cells. The researchers checked whether their transplanted tissue induced cardiac arrhythmias in the mice – a danger when introducing immature cells into the damaged heart — and they did not.
Yoon says it took almost 9 years to complete the project; an important next step is to test direct reprogramming with human cells.
This work was supported by grants from the National Heart Lung and Blood Institute (R01HL150877, R61HL 154116, R01HL125391) and a American Heart Association Transformative Project Award.