Alcohol exposure is known to perturb fetal heart development; half of all children with fetal alcohol syndrome have congenital heart defects, such as arrhythmias or structural abnormalities. Chunhui Xu and colleagues recently published a paper in Toxicological Scienceson how human cardiac muscle cells, derived from iPS (induced pluripotent stem cells), can be used as a model for studying the effects of alcohol.
Alcohol-induced cardiac toxicity is usually studied in animal models, but human cells are different, and a cell-culture based approach could make it easier to study the effects of alcohol and possible interventions more easily.
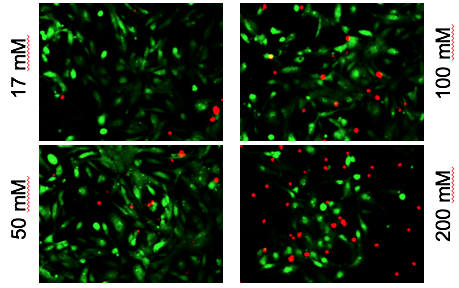
Red shows toxic effects of alcohol on iPS-derived cardiomyocytes
Xu and her colleagues observed that high levels of alcohol damaged cardiac muscle cells and put them under oxidative stress. But even at relatively low concentrations of alcohol, the researchers also saw perturbations in cells’ electrical activity and the ability to contract, which reasonably matches the effects of alcohol on human heart development. The lowest level tested was 17 millimolar – the legal limit for driving in most states (0.08% blood alcohol content). Read more





