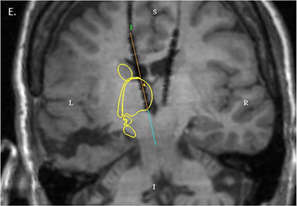
Emory neurosurgeon Robert Gross was recently quoted in a Tennessee newspaper article about a clinical trial of cell therapy for stroke. He used cautionary language to set expectations.
“We’re still in the very early exploratory phases of this type of work,” Gross told the Chattanooga Times Free Press. “In these cases, a significant area of the brain has been damaged, and simply putting a deposit of undifferentiated cells into the brain and magically thinking they will rewire the brain as good as new is naive. None of us think that.”
A more preliminary study (just 18 patients) using the same approach at Stanford and University of Pittsburgh was published this summer in Stroke, which says it was the “first reported intracerebral stem cell transplant study for stroke in North America.” The San Diego Union Tribune made an effort to be balanced in how the results were described:
Stroke patients who received genetically modified stem cells significantly recovered their mobility… Outcomes varied, but more than a third experienced significant benefit.
The newspaper articles made us curious about what these cells actually are. They’re mesenchymal stromal cells, engineered with an extra modified Notch gene. That extra gene drives them to make more supportive factors for neurons, but it doesn’t turn them into neurons. Read more