In the 1999 film The Matrix, the character Neo is offered a choice between a blue pill (to forget) and a red pill (to remember). If only neuroscience was that simple! It may be that neurons need both red and blue, possibly an elaborate dance of molecules, for a fragile memory to lodge itself in the brain.
Neuroscientists Kimberly Maguschak and Kerry Ressler provide a glimpse into this process with their recent paper in the Journal of Neuroscience.
Ressler is both a psychiatrist and a Howard Hughes Medical Institute-supported researcher with a laboratory at Yerkes National Primate Research Center. Maguschak completed her doctorate at Emory and is now a postdoc with Guoping Feng at MIT.
The research is a follow-up on their work probing the role of beta-catenin in fear memory formation. We previously described this protein as acting “like a Velcro strapâ€, attaching cells’ internal skeletons to proteins on their external membranes that help them adhere to other cells. If brain cells need to change shape and form new connections for memories to be consolidated, we can see how this kind of molecule would be important.
Beta-catenin is also central to a signaling circuit that maintains stem cells and prods an embryo to separate into front and back or top and bottom. This circuit is called “Wnt†(the name is a fusion of the fruit fly gene wingless and a cancer-promoting gene discovered in mice, originally called Int-1).
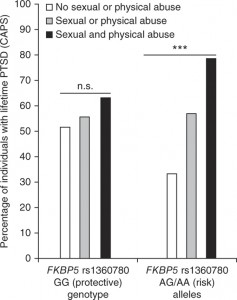
Maguschak and Ressler wanted to assess the role Wnt signals play in learning and memory. The model system was the same as in their previous work: if mice are electrically shocked just after they hear a certain tone, they gradually learn to fear that tone, and they show that fear by freezing.

Kerry Ressler, MD, PhD
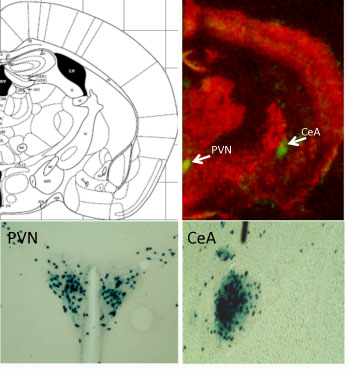
Maguschak saw that in the amygdala, a part of the brain important for fear responses, Wnt genes are turned down during the learning process temporarily but then come back on. If the mice only hear the tone or only get the shock, the genes’ activities don’t change significantly.
She then introduced proteins that perturb Wnt signaling directly into the amygdala. Extra Wnt injected before training, while it didn’t stop the mice from learning to fear the tone, made that training less likely to “stick.†Two days later, the mice that received Wnt didn’t seem to fear the tone as much.
Here’s the possibly confusing part: a Wnt inhibitor also impaired fear memory consolidation. In effect, both blue and red pills actually interfered with how well memories endured. The authors suggest this is because Wnt signals have to be turned down during fear memory formation but then turned back up so those memories can solidify. The Wnt signals seem to go along with the adhesive interactions of beta-catenin. It looks like beta-catenin’s stickiness also needs to be tuned down and then back up.
The off-then-on-again requirement Maguschak and Ressler observe is reminiscent of results from cell biologist James Zheng’s lab. He and his colleagues saw that the actin cytoskeleton needed to be weakened and then stabilized during long-term potentiation, an enhancement of connections between neurons thought to lie behind learning and memory.
Several laboratories have identified potential drugs that modify beta-catenin/Wnt. These new results suggest that the timing of when and how to use such drugs to enhance memory may critically important to consider, Ressler says.
“To interfere with memory formation after trauma or enhance memory formation in people with dementia, researchers will clearly need to attend to the full complexity of the dynamics of synaptic plasticity and memory,” he says.
A nifty link to an animation of Wnt signaling