Biochemists at Emory are achieving insights into how an important regulator of the immune system switches its function, based on its orientation and local environment. New research demonstrates that the glucocorticoid receptor (or GR) forms droplets or “condensates” that change form, depending on its available partners.
The inside of a cell is like a crowded nightclub or party, with enzymes and other proteins searching out prospective partners. The GR is particularly well-connected and promiscuous, and has the potential to interact with many other proteins. It is a type of protein known as a transcription factor, which turns some genes on and others off, depending on how it is binding DNA.
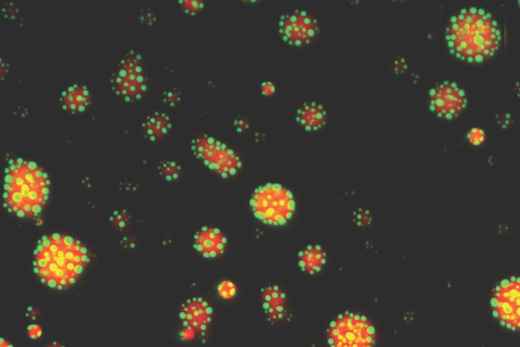
“It is now thought that most transcription factors form or are recruited into condensates, and that condensation modulates their function,” says Filipp Frank, PhD, first author of the paper and a postdoctoral instructor in Eric Ortlund’s lab in the Department of Biochemistry. “What’s new is that we identified a DNA-dependent change in GR condensates, which has not been described for other transcription factors.”
The results are published in Proceedings of the National Academy of Sciences. Ortlund is a co-author of the paper, along with postdoctoral fellow Xu Liu, PhD.
Understanding how the GR works could help researchers find anti-inflammatory drugs with reduced side effects. The GR is the target for corticosteroid drugs such as dexamethasone, which is currently used to treat COVID-19 as well as allergies, asthma and autoimmune diseases.
Corticosteroids’ harmful side effects are thought to come from turning on genes involved in metabolism and bone growth, while their desired anti-inflammatory effects result from turning other inflammatory and immune system genes off. Researchers want to find alternatives that could separate those two functions.
In the laboratory, GR’s reversible condensation can be glimpsed without a microscope or other equipment. In the video, a tube of a solution of GR protein warms up and the solution turns cloudy, because phase separation of the droplets is occurring. When the tube is returned to an ice bucket, the solution is clear again.
Also, solutions of the protein tend to form strings when extruded out of a thin opening, resembling a nylon synthesis reaction.
What the Emory researchers have observed is that GR’s condensation depends on how it is binding DNA. At some sites, it pairs up or “dimerizes” – turning genes on. At others, it binds one at a time, turning genes off. The two types of condensates do not mix, and they bring different sets of other proteins called co-regulators to the party depending on how they are binding DNA. Various parts of the GR protein, including the module that contacts DNA and the module that binds hormones or drugs, are necessary for condensate formation with different partners.
“Recruitment of co-regulators to DNA is a critical function of GR for the activation of specific genes,” Frank says. “We are proposing that GR’s interaction with different types of DNA sequences controls the recruitment of co-regulators by changing the properties of GR droplets.”
The research was supported by the W.M. Keck Foundation, the National Institute of Diabetes and Digestive and Kidney Diseases (R01DK115213) and a Winship Cancer Institute pilot grant.

