Scientists have revealed molecular differences between how the African and Asian strains of Zika virus infect neural progenitor cells. The results could provide insights into the Zika virus’ recent emergence as a global health emergency, and also point to inhibitors of the p53 pathway as potential leads for drugs that could protect brain cells from cell death.
The findings, from the Emory/Johns Hopkins/Florida State team that showed this spring that neural progenitor cells are particularly vulnerable to Zika infection (related paper), were published this week in Nucleic Acid Research. The manuscript was also posted on BioRxiv before publication.
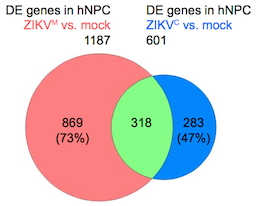
Overlap in gene expression changes when neural progenitor cells are infected by African or Asian strains of Zika virus. Diagram from Nucleic Acids Research via Creative Commons.
Zika virus was first discovered in Uganda in the 1940s, and two distinct lineages of Zika diverged sometime in the second half of the 20th century: African and Asian. The strains currently circulating in the Western Hemisphere, which have been linked to microcephaly in infants and Guillain-Barre syndrome in adults, are more closely related to the Asian lineage.
The research team catalogued and compared genes turned on and off by Asian and African strains of Zika virus, as well as dengue virus, in human neural progenitor cells. The authors describe dengue as inducing more robust changes in gene expression than either strain of Zika. Although they show that dengue can infect neural progenitor cells like Zika can, dengue infection does not stunt the cells’ growth or lead to cell death.
“This shows that the differences between Zika and dengue are not at the level of being able to infect neural progenitors, but more about the harm Zika causes when it does infect those cells,” says senior author Peng Jin, PhD, professor of human genetics at Emory University School of Medicine. Read more