How often can doctors go from encountering a patient with a mysterious disease, to finding a mutation in a gene that causes that disease, to developing a treatment crafted for that mutation?
This is true personalized molecular medicine, but it’s quite rare.
How rare this is, I’d like to explore more, but first I should explain the basics.
At Emory, Stephen Traynelis and Hongjie Yuan have been working with Tyler Pierson, David Adams, William Gahl, Cornelius Boerkoel and doctors at the National Institutes of Health’s Undiagnosed Diseases Program (UDP) to investigate the effects of mutations in the GRIN2A gene.
Their report on the molecular effects of one such mutation, which caused early-onset epilepsy and intractable seizures in a UDP patient, was recently published in Nature Communications.
With that information in hand, UDP investigators were able to repurpose an Alzheimer’s medication as an anticonvulsant that was effective in reducing seizure frequency in that patient. [The details on that are still unpublished but coming soon.]
The GRIN2A gene encodes one of several NMDA receptor subunits, important signaling molecules in the brain. Traynelis and his colleagues are well-versed in the http://www.troakley.com/ biology of NMDA receptors, so it was natural that the UDP researchers came to them, after first identifying the GRIN2A mutation through whole-exome sequencing of the patient and his family.
From the Results section, here’s a summary of how severe this case was. Imagine what his parents have been going through.
A six-year-old boy [he is now nine or ten] was admitted to UDP with a history of intractable infantile-onset epilepsy and profound global developmental delay with no attainment of any milestones, not even head control. At presentation, he was having daily, generalized seizures that had been refractory to multiple anti-convulsants…
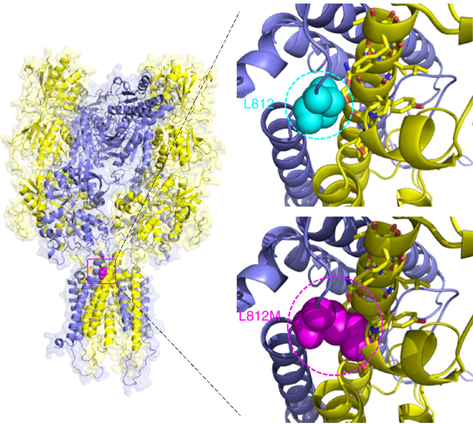
A model showing the effect of changing one amino acid in the NMDA receptor. The mutation is in a region that appears to control the receptor’s open-and-close gating function.
In the laboratory, Yuan and Traynelis discovered that the mutation in the GRIN2A gene dramatically overactivated the NMDA receptor molecule. This in turn probably was driving enhanced neuronal excitability, leading to the seizures. Traynelis says that the boy’s mutation changed only one amino cheap oakley acid in the protein, and it wasn’t clear simply from looking at the change what the effect was.
“We might see seizures and neurodegeneration, and think that the mutation is overactive, but it’s simply speculation until we do the experiment,†he says.
Going further, investigators were able to use the specific information about how the mutation changed the receptor to suggest a potential treatment: memantine.
Memantine, a known NMDA receptor antagonist, is used clinically as a treatment for the symptoms of Alzheimer’s disease. While it has been shown to have anticonvulsant properties in most animal models of epilepsy, it has not been used extensively for that purpose in humans. Some limited studies of memantine have been performed in children with autism cheap oakley sunglasses spectrum disorders.


