Guest post from Courtney St Clair Ardita, MMG graduate student and co-author of the paper described. Happy Halloween!
In the past, reactive oxygen species were viewed as harmful byproducts of breathing oxygen, something that aerobic organisms just have to cope with to survive. Not any more. Scientists have been finding situations in humans and animals where cells create reactive oxygen species (ROS) as signals that play important parts in keeping the body healthy.
One example is when commensal or good bacteria in the gut cause the cells that line the inside of the intestines to produce ROS. Here, ROS production helps repair wounds in the intestinal lining and keeps the environment in the gut healthy. This phenomenon is not unique to human intestines. It occurs in organisms as primitive as fruit flies and nematodes, so it could be an evolutionarily ancient response. Examples of deliberately created and beneficial ROS can also be found in plants, sea urchins and amoebas.
Researchers led by Emory pathologist Andrew Neish have taken these findings a step further and identified the cellular components responsible for producing ROS upon encountering bacteria. Postdoctoral fellow Rheinallt Jones is first author on the paper that was recently published in The EMBO Journal.
Using a special dye that fluoresces when it comes in Ray Ban outlet contact with ROS, the researchers were able to show that ROS is produced in fruit fly intestines after they are fed different strains of bacteria. The researchers also demonstrated that ROS production occurs in mouse intestine as well. The group found that, of all the commensal bacteria they tested, Lactobacillus strains seemed to induce the most ROS. L. plantarum and L. rhamnosus, bacteria found naturally in fly and mouse intestines, respectively, produced the most ROS in each model. Thus, epithelial cells have the strongest response to beneficial types of bacteria known to be present in the intestine. No ROS production occurred when cells were exposed to pathogenic Salmonella bacteria, so the ability to stimulate ROS production seems to be unique to commensal bacteria.
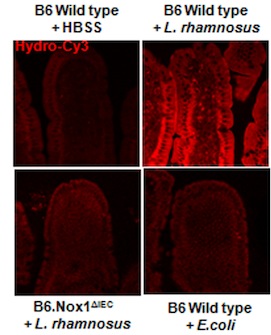
Hydro-Cy3 is a dye that fluoresces when it comes into contact with reactive oxygen species. ROS is produced when mouse intestines are treated with Lactobacillus rhamnosus but not when they are treated with buffer or E coli. Intestines from mice that lack the Nox1 enzyme do not produce ROS after L. rhamnosus treatment.
An enzyme known as Nox1 is responsible for creating the ROS in the intestine. Nox1 is related to Nox2, the enzyme that the immune system uses to generate bactericidal ROS in some types of white blood cells. However, the Nox1-generated ROS are not a weapon of defense; instead, they form a message of cooperation.
With the help of genetically altered animals, the researchers found that intestinal epithelial cells of flies and mice that lacked the gene for Nox1 did not release ROS after bacterial stimulation.  Nox1 was necessary for intestinal epithelial cells to release ROS. Nox1’s ability to produce ROS after bacterial stimulation must be evolutionarily ancient since the enzyme is serving a similar role in organisms as distantly related as flies and mice.
Bacteria are often thought of as disease-causing agents that need to be avoided. Yet, science is proving again and again that this way of thinking is quite narrow-minded. This new finding highlights the possibility that organisms have been relying on commensal bacteria and ROS together to stay healthy for millions of years. The identification of Nox1 as the cellular component that is involved in ROS production after bacterial stimulation helps scientists further understand the probiotic process and may lead to the development of new treatments for intestinal diseases. By looking for bacterial components that stimulate Nox1, scientists could identify molecules that could be as drugs to promote intestinal healing.

