How can we study depression and antidepressants in animals? They can’t talk and tell us how they’re feeling. Previously, researchers have used the model of “behavioral despair,†with examples of the forced swimming test or the tail suspension test.

Shannon Gourley, PhD
Several psychiatrists have been arguing that a new framework is needed, which better simulates aspects of depression in humans, such as the variety of behavioral changes and the several week time period needed for antidepressants to function. This new framework could help illuminate how depression develops, and lead to new antidepressants that are effective for more people.
Shannon Gourley, who recently joined the Emory-Children’s Pediatric Research Center has been taking the approach of examining the lack of motivation and self-defeating behavior that are integral parts of depression.
The Pediatric Research Center is an effort led by Emory University and Children’s Healthcare of Atlanta, including partnerships with the Georgia Institute of Technology and Morehouse School of Medicine.
Note: Gretchen Neigh in psychiatry/physiology has been doing work with a similar theme, looking at the effects of adolescent social stress in animal models.
Gourley, neuroscience graduate student Andrew Swanson and their colleagues at Yale, where Gourley was a postdoc with Jane Taylor and Tony Koleske, have a new paper in PNAS on this topic. In particular, they dissect how chronic stress – or exposure to the stress hormone corticosterone – can produce loss of motivation and impaired decision making.
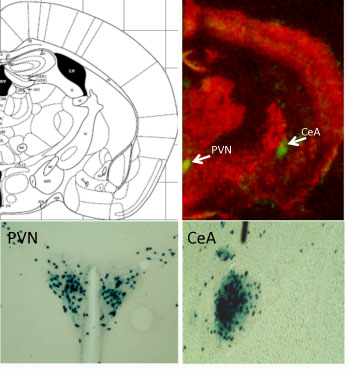
First, the researchers found that exposing rodents to cheap oakleys corticosterone shut off a growth factor called BDNF (brain-derived neurotrophic factor) in the frontal cortex, a region of the brain important for planning and goal-directed behavior. BDNF nourishes neurons and helps keep them alive.
To confirm that BDNF was important in this region of the brain, researchers selectively silenced the gene for BDNF only in the frontal cortex. Both mice exposed to stress hormones and the BDNF-altered mice showed reduced motivation to earn food rewards. Mice would ordinarily press a lever dozens of times to get a food pellet, but the BDNF-altered animals would stop trying earlier – the “break point†is 2/3 as high.
“Depression is a leading cause of unemployment because people are unable to break out of self-defeating behavioral patterns and to muster the motivation to engage with the world. If we can better understand how to treat these symptoms, we can effect better outcomes for individuals suffering from depression,†Gourley says. “The BDNF deficiency alone could account for the loss of motivation that individuals with depression suffer.â€
However, she reports her team was surprised that the loss of BDNF could not account for another aspect of depression: cyclical self-defeating behavior. They modeled this by asking whether mice continue to press a lever for a food reward even when the reward is no longer available.
“When we made the discovery that reduced BDNF could not account for all of the depression symptoms that we study, we took a step back and looked at the stress response system,†Gourley says.
Stress hormone exposure impairs the ability of mice to switch away from fruitless behaviors, but loss of BDNF in the frontal cortex does not. Here, the stress response system itself was the culprit. When her team temporarily blocked the ability of mice to shut off their stress response systems using the drug mifepristone, mice had impaired decision-making. However, their motivation to obtain rewards was not altered. When the drug wore off, they returned to normal.
Gourley says the implication is that effective antidepressants need to be able to attack not one, but two physiological systems: they need to increase levels of BDNF, and they need to help the stress system recover so that it can shut itself off better. A classic trycyclic antidepressant, amitriptyline, can do both and was effective in treating both the motivation and decision making parts of depression in animal models.
The use of tricyclic antidepressants is limited because of side effects and overdose potential. In addition, another challenge in treating depression is that current antidepressants only begin to work after several weeks or months of treatment. This is thought to be because it takes several weeks for these drugs—which act only indirectly on BDNF—to restore BDNF levels back to normal.
New compounds that act directly on BDNF’s receptor TrkB, such as those identified and tested by Emory researcher Keqiang Ye, could be promising in the development of new approaches to depression, Gourley says.
She and her team also showed that a drug called riluzole, which acts indirectly but rapidly on BDNF systems, has antidepressant effects in the animal models. Riluzole is currently in use to treat ALS, and reportedly has antidepressant effects in humans. Clinical trials with riluzole in the context of depression are underway.







 Larry Young is quoted extensively in
Larry Young is quoted extensively in